Downstream Packaging Line Design in Pharmaceutical Production: Optimize Efficiency and Quality
Introduction: Why Downstream Design Determines Line Success
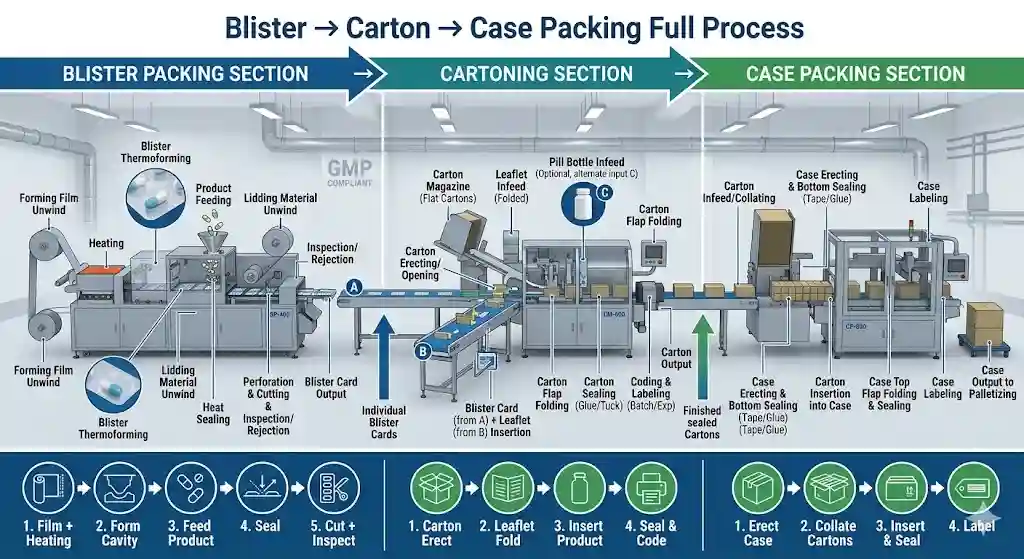
In pharmaceutical manufacturing, the downstream packaging line is often the true bottleneck determining throughput and product quality. Even a high-precision blister packaging machine can underperform if cartoning, case box packaging, or line layout aren't optimized.
In our own experience of maintenance: Many production issues, from jams to damaged packs, stem from poor integration rather than faulty machines.
Core Components of Downstream Line Design
1. Blister-to-Carton Transfer
- Ensures blister packs are accurately fed into cartons using gentle conveyors, pushers, or robotic arms.
- Sensors detect pack positions to prevent misalignment.
- Key consideration: speed synchronization to avoid line stoppages.
- Practical note: High-speed lines often require 1–2 buffer zones to handle minor jams without halting the line.
2. Cartoning Machines
- Folds, fills, and closes cartons efficiently, handling various blister formats.
- Inline inspection systems check carton integrity and reject defective packs.
- Real-use tip: Misaligned blisters are one of the top causes of downtime in pharmaceutical lines.
3. Case Packing & Palletizing
- Groups cartons into cases for shipping with precise spacing, orientation, and stacking.
- Automated palletizers improve efficiency on high-volume lines.
- Ergonomics and operator safety must be considered, especially in semi-automated areas.
4. Integration & Line Synchronization
- Real-time communication between machines via PLC or SCADA ensures smooth operation.
- Buffer zones prevent minor issues from stopping the entire line.
- Continuous monitoring allows dynamic speed adjustment and error handling.
- Engineer’s note: Even small misalignments can cascade into larger downtime if buffers or sensors aren’t properly configured.
Common Design Challenges
- Line imbalance: blister machine output exceeds cartoning capacity.
- Product damage: high-speed transfers or misaligned cartons causing broken blisters.
- Downtime propagation: one jam halting the entire line if buffering is insufficient.
- Flexibility vs efficiency: multi-SKU lines require modular, adjustable setups.
Best Practices for Engineers
- Map the Complete Workflow: Include all transfer points and inspection stations.
- Implement Real-Time Feedback: Sensors and PLCs adjust speeds dynamically.
- Design for Flexibility: Accommodate multiple pack sizes and quick SKU changeovers.
- Prioritize Gentle Handling: Reduce mechanical stress on blisters to avoid deformation or sealing failures.
- Routine Validation & Maintenance: Test the entire line under production conditions.
Applications
Optimized downstream lines are essential in:
- Pharmaceutical tablet and capsule production
- OTC drug manufacturing
- Nutraceutical and supplement packaging
FAQ
Q1: Why is downstream line design critical for pharma production?
Even high-precision machines can underperform if transfer, cartoning, or case packing is inefficient.
Q2: How do engineers prevent line stoppages?
Using buffer zones, synchronized machine speeds, and real-time sensors to manage flow.
Q3: Can the same downstream line handle multiple SKUs?
Yes, with flexible conveyors, adjustable cartoning stations, and modular case packing setups.
Optimize Your Downstream Packaging Today
A high-performing blister packaging line is only as effective as its downstream design.
If your line suffers from jams, misaligned packs, or inconsistent throughput:
Our engineers can help you:
• Map and optimize your downstream workflow
• Improve line synchronization and buffer design
• Minimize product damage and downtime
• Ensure regulatory compliance
More info may you interesting:



